Galvanic series anode selection is the engineering process of choosing a sacrificial metal with a more negative electrochemical potential than the protected structure. By referencing the galvanic series, engineers select anodes—typically Zinc, Aluminum, or Magnesium—that provide a driving voltage (usually >0.25V difference) to mitigate corrosion without inducing hydrogen embrittlement.
Corrosion is a relentless chemical tax on global infrastructure. For Corrosion Engineers and Marine Architects, the difference between a 20-year asset life and a catastrophic failure often comes down to a few millivolts on the galvanic series. Selecting the right anode isn't just about picking a "sacrificial" metal; it's about managing the electrochemical dance between the electrolyte and the submerged surface.
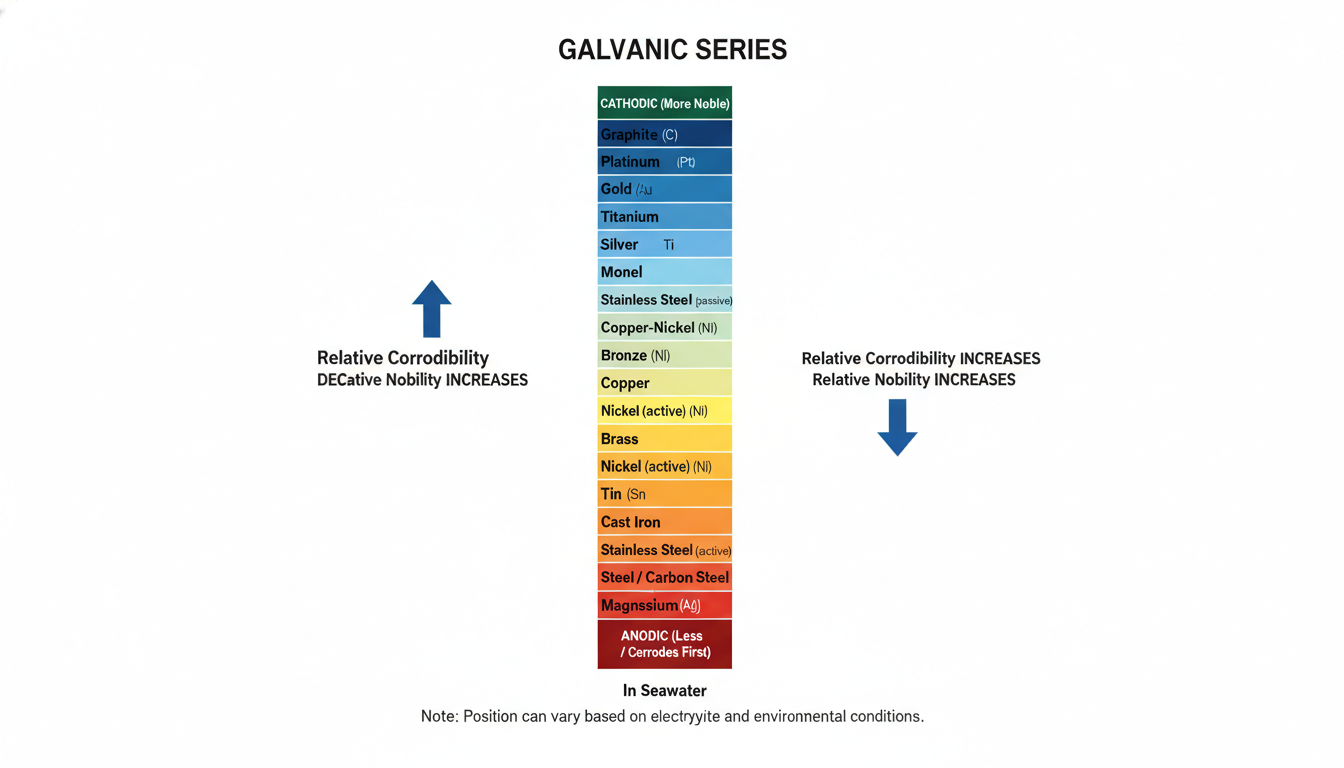
The Galvanic Series is a hierarchy of metals ranked by their potential in a specific electrolyte, usually seawater. Unlike the Standard Electromotive Force (EMF) series, which uses pure solutions, the galvanic series reflects real-world conditions. This distinction is vital: a metal’s position can shift based on the salinity and aeration of the water.
Historically, the work of Francis LaQue at the International Nickel Company established the foundation of modern marine corrosion data. Today, organizations like AMPP (formerly NACE International and SSPC) maintain these charts to reflect modern alloys. The Anodic Index provides the relative voltage difference required to predict if two metals will experience galvanic corrosion when coupled.
 Sacrificial Anode Materials: Zinc, Aluminum, and Magnesium
Sacrificial Anode Materials: Zinc, Aluminum, and MagnesiumChoosing a Sacrificial Anode requires balancing electrochemical capacity, driving voltage, and material cost. While Zinc was the historical standard, modern engineering leans heavily toward optimized alloys.
Zinc (MIL-A-18001K): The traditional choice for salt water. It is reliable but heavy and has a lower "capacity" (Amp-hours per pound) than aluminum.
Indium-activated Aluminum: These are the current industry favorites. Aluminum is lighter and offers a higher electrochemical capacity. The addition of Indium prevents the formation of a tight oxide film (Passivation), ensuring the anode remains active.
Magnesium: Boasting the highest driving voltage, Magnesium is reserved for high-resistivity environments like fresh water. Using it in salt water usually leads to "over-protection" and rapid consumption.
A critical concern for modern offshore projects is the interaction between anodes and Super-duplex stainless steels. If the driving voltage is too high, you risk hydrogen-induced stress cracking (HISC). We found that using traditional Zinc can sometimes provide a "safer" potential floor than high-output Aluminum for these specific sensitive alloys.
The environment dictates anode efficiency. In Brackish water, the high Resistivity of the electrolyte limits current throw. This is where Environmental Nuance becomes a design factor. If you use a Zinc anode in a river estuary where salinity fluctuates, the anode may develop a hard crust of zinc carbonate, effectively "turning off" the protection.
High-temperature applications, such as heat exchangers or hot water storage tanks, present a unique risk. Above 140°F (60°C), Zinc can undergo a "potential reversal," where it becomes noble to the steel it is supposed to protect. In these cases, we always specify high-temperature Aluminum alloys or Magnesium, depending on the fluid chemistry.
Anode Sizing Calculations are the backbone of a professional Cathodic Protection (CP) plan. You cannot simply "guess" the number of anodes. The design must account for the Current Density required to polarize the structure and the Design Life of the system.
The fundamental formula for determining the total mass of anode material required is:
W = (I × L × 8760) / (u × C)
Where:
W: Total weight of anode material (kg)
I: Total current required (Amps)
L: Design life (years)
8760: Hours in a year
u: Utilization factor (usually 0.8 to 0.9)
C: Electrochemical capacity of the alloy (Ah/kg)
Using Ohm's Law (V=IR), we also calculate the resistance of the anode to ensure it can actually deliver the required current at the end of its life when its surface area has diminished.
Compliance is non-negotiable in industrial and marine sectors. For the US Military and defense contractors, MIL-STD-889 provides the definitive guide for galvanic compatibility. In the commercial marine world, ABYC E-2 governs cathodic protection for small craft, while ISO 12473 and NACE SP0169 are the bibles for global offshore and pipeline engineering.
For Boat Owners and field technicians, a Visual Decision Tree simplifies the Selection Logic. The process follows a strict hierarchy of Electrolyte > Structural Material > Temperature.
If the electrolyte is fresh water (resistivity > 10,000 ohm-cm), Magnesium is the only viable choice. If the water is salt (resistivity < 50 ohm-cm), Aluminum is the most cost-effective and efficient solution for 90% of applications.
The Port of Savannah Piling Failure (2019)
A recent project led by William Drake involved a series of steel pilings that showed 30% section loss within just five years, despite having Zinc anodes installed. Upon investigation, our team, collaborating with insights from Julie Holmquist of Cortec Corporation, discovered that the "zinc" anodes were actually a low-grade alloy with high iron impurities.
The iron impurities caused the anodes to form a non-conductive layer. We replaced the system with Indium-activated Aluminum anodes and implemented a Vapor phase Corrosion Inhibitor (VpCI) for the splash zone. Results? The polarization potential stabilized at -910 mV (CSE) within 48 hours, meeting NACE standards perfectly. Experts like Dominique Thierry and Luke Stone have long argued that material purity is as important as the galvanic series position itself.
In most marine environments, anodes are designed for a 1-to-3-year lifespan. However, in industrial CP systems for pipelines, 20-year lifespans are common, achieved through massive anode sleds and precise current calculations.
It is generally discouraged. Because Aluminum has a slightly more negative potential than Zinc, the Aluminum will often "protect" the Zinc anode, causing the Aluminum to deplete faster while the Zinc remains largely intact. Stick to one alloy per system.
To check if your anodes are working, use a high-impedance multimeter and a Silver/Silver Chloride (Ag/AgCl) reference electrode. For a steel hull in seawater, you are looking for a reading more negative than -800 mV.