The Evolution of Spinal Instrumentation: Why Grade 23 ELI Titanium Matters
Spinal surgery has transitioned from rigid stabilization to dynamic biological integration. In this shift, titanium spinal rods have emerged as the backbone of modern orthopedic intervention.
While Grade 5 titanium was once the primary choice, 2026 clinical data favors Grade 23 ELI Titanium. ELI stands for "Extra Low Interstitials," referring to the reduced levels of oxygen, nitrogen, and iron within the alloy.
This high-purity composition, strictly governed by ASTM F136 compliance, provides the enhanced ductility required for complex rod contouring without sacrificing tensile strength. For surgeons, this means a lower risk of intraoperative rod breakage.
Understanding the Mechanical Edge: Elastic Modulus and Implant Stability
Titanium Grade 23 ELI offers an elastic modulus of approximately 105-120 GPa. This specific range allows the implant to share physiological loads with the surrounding bone, effectively reducing the risk of implant loosening and adjacent segment disease.
A major challenge in spinal fusion is "stress shielding." When an implant is too stiff, it carries all the mechanical load, causing the surrounding bone to weaken and resorb. Stainless steel rods have a modulus of roughly 200 GPa—nearly double that of titanium.
By using a material that sits closer to human cortical bone (10-30 GPa), we significantly lower the implant loosening risk. This mechanical harmony ensures mechanical stability in vivo over decades, not just years.
The Bio-Mechanical Synergy Protocol: A Framework for Material Selection
At China Titanium Factory, we utilize the Bio-Mechanical Synergy Protocol (BMSP) to vet all medical grade titanium sourcing. This proprietary framework evaluates four critical pillars:
Fatigue Mapping: Measuring the rod's endurance limit under millions of loading cycles.
Ductility Calibration: Ensuring the rod can be bent to the patient's spinal curvature without creating micro-fractures.
Morphological Integrity: Analyzing grain structure to prevent premature fatigue failure.
Surface Passivation: Confirming the presence of a stable titanium oxide layer for biocompatibility.

Osseointegration Properties and Long-Term Clinical Success
One of the primary reasons Grade 23 ELI Titanium is considered the gold standard is its Osseointegration Properties. The material is biologically inert, meaning it does not trigger an adverse immune response.
"Osseointegration is the functional and structural connection between living bone and the surface of a load-bearing implant." —National Institutes of Health
The Ti-6Al-4V ELI composition promotes the formation of a stable titanium dioxide layer. This layer serves as a scaffold for bone cells to attach and grow directly onto the rod surface, providing secondary stability that prevents long-term hardware migration.
Manufacturing Excellence: From ASTM F136 to Additive Manufacturing
Traditional spinal rod fabrication involves precision machining and cold-working to achieve high fatigue life. However, as we move through 2026, additive manufacturing (3D printing) is revolutionizing custom spinal solutions.
3D-printed Grade 23 ELI allows for porous structures that further enhance bone ingrowth. Whether through traditional or additive methods, adherence to ASTM F136 compliance is non-negotiable to prevent catastrophic failure.
| Property | Grade 5 (Ti-6Al-4V) | Grade 23 (ELI) |
|---|---|---|
| Oxygen Content | Max 0.20% | Max 0.13% |
| Ductility | Moderate | Superior |
| Fracture Toughness | High | Excellent |
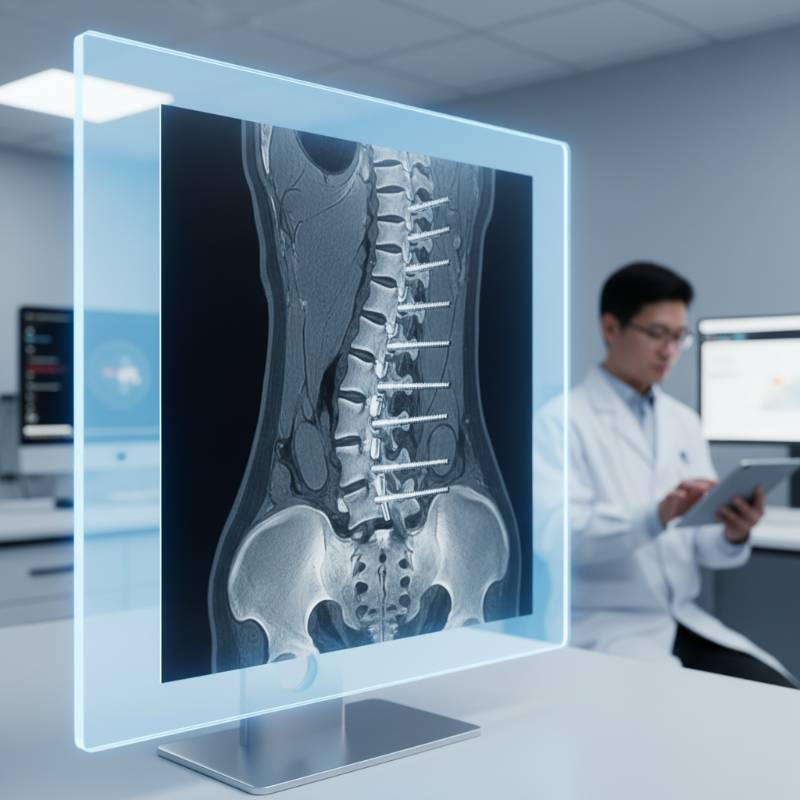
Imaging and Patient Safety: Is Grade 5 or Grade 23 Titanium MRI Safe?
A common question from patients and clinicians is: Is Grade 5 titanium rod MRI safe? The answer is a definitive yes. Titanium is non-ferromagnetic, meaning it is not attracted to magnetic fields.
In practice, this means titanium rods will not shift or heat up during a scan. Furthermore, titanium produces significantly fewer image artifacts compared to stainless steel or cobalt-chrome. This allows for clear CT and MRI diagnostic safety, enabling surgeons to monitor the fusion site with high precision.
Sustainability and Economic Impact in Medical Device Procurement
The 2026 healthcare market is increasingly focused on the carbon footprint of medical devices. While titanium ore processing is energy-intensive, the longevity of Grade 23 ELI Titanium offers a "Revision-Reduction Dividend."
By reducing the rates of implant failure and subsequent revision surgeries, the healthcare system saves massive amounts of energy and resources. High-durability materials are the most sustainable choice because they are "one-and-done" solutions for the patient.
Frequently Asked Questions About Titanium Spinal Rods
Why is ELI preferred over standard Grade 5 for spinal rods?
Grade 23 ELI has lower oxygen content, which significantly improves ductility and fracture toughness. This makes the rods easier to contour during surgery without risking micro-cracks that could lead to fatigue failure.
Does titanium interfere with postoperative CT scans?
Titanium is the preferred material for imaging. While all metals create some "starburst" effect, titanium's lower atomic number results in minimal artifacts, allowing for better visualization of the spinal canal.
How long can a Grade 23 ELI rod last in the body?
Designed for permanent implantation, these rods are engineered to last the patient's lifetime. Their high fatigue resistance and biocompatibility ensure they remain stable as long as the fusion is maintained.